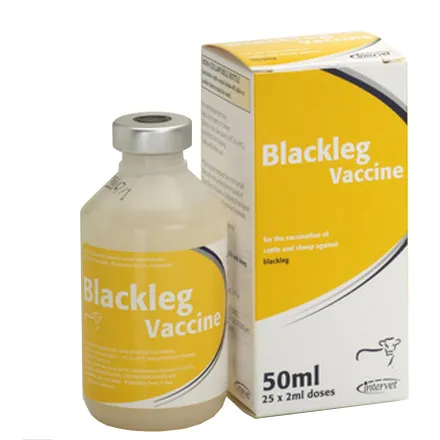
Blackleg Injection 50ml
$28.79
$57.01
Click here to Download Data Sheet Summary of Product Characteristics 1 NAME OF THE VETERINARY MEDICINAL PRODUCT BLACKLEG VACCINE 2 QUALITATIVE AND QUANTITATIVE COMPOSITION For a full list of excipients, see section 6.1. 3 PHARMACEUTICAL FORM Suspension for injection 4 CLINICAL PARTICULARS 4.1 Target Species Sheep and cattle 4.2 Indications for use, specifying the target species For the active immunisation of sheep and cattle to reduce mortality and clinical signs of blackleg disease caused by Clostridium chauvoei. 4.3 Contraindications None. Active substance(s): per dose (2 ml) Cl.chauvoei - strain No: 655 2.50 x 108 cells & equivalent toxoid 656 2.50 x 108 cells & equivalent toxoid 657 2.50 x 108 cells & equivalent toxoid 658 2.50 x 108 cells & equivalent toxoid 1048 2.50 x 108 cells & equivalent toxoid Adjuvant(s): Aluminium hydroxide 400 mg Preservative(s): Thiomersal 0.26 mg 4.4 Special warnings for each target species Do not vaccinate unhealthy animals. When vaccinating pregnant animals, stress should be avoided particularly during the later stage of pregnancy. 4.5 Special precautions for use Special precautions for use in animals None. Special precautions to be taken by the person administering the veterinary medicinal product to animals In the case of accidental self injection or ingestion seek medical advice immediately and show the package leaflet or the label to the physician. 4.6 Adverse reactions (frequency and seriousness) Vaccination of lambs may result in temporary swellings at the injection site lasting for up to 3-4 months after vaccination. Typically, these swellings may be warm when compared to the surrounding area for up to 14 days after vaccination. Safety studies in lambs have shown that the swellings did not appear to inconvenience the animals or hinder neck movement. Minor temperature increases (approximately 1°C – 2°C) lasting for up to 1 week may occur following vaccination of lambs. No local or systemic reactions have been reported when vaccinating calves. Occasional hypersensitivity reactions may occur. 4.7 Use during pregnancy, lactation or lay The vaccine may be administered to pregnant sheep provided dosing is completed 4 –6 weeks prior to predicted lambing date. The vaccine cannot be administered to pregnant cows. 4.8 Interaction with other medicinal products and other forms of interaction No information is available on the safety and efficacy from the concurrent use of this vaccine with any other. It is therefore recommended that no other vaccines should be administered within 14 days before or after vaccination with Blackleg Vaccine. 4.9 Amounts to be administered and administration route Administration is by subcutaneous injection. Sheep: Basic vaccination Two injections, each of 2ml, separated by an interval of 4-6 weeks to be completed 2-3 weeks before onset of the period of risk. Minimum age of vaccination is 3 weeks. Re-vaccination scheme Thereafter a 2ml booster injection at intervals of not more than 12 months. Cattle: Basic vaccination Two injections, each of 2ml, separated by an interval of 4-6 weeks to be completed 2-3 weeks before onset of the period of risk. Minimum age of vaccination is 4 weeks. Re-vaccination scheme Thereafter a 2ml booster injection at intervals of not more than 12 months. Shake vaccine bottle well before use. Do not freeze. Syringes and needles should be from gamma irradiated packs or freshly sterilised by boiling for 20 minutes. No alcohol or disinfectant should be used for this sterilisation procedure. The use of an automatic vaccinator is recommended. Since the bottle is non-collapsible, a vaccinator with a vented draw-off spike or similar device must be used. The instructions supplied with such syringes should be noted and care should be taken to ensure the delivery of the full dose, particularly with the final few doses from the bottle. 4.10 Overdose (symptoms, emergency procedures, antidotes), if necessary Accidental overdosage is unlikely to cause any reaction other than described in point 4.6. 4.11 Withdrawal Period(s) Zero days. 5 PHARMACOLOGICAL or IMMUNOLOGICAL PROPERTIES ATC vet code: QI02AB01; Pharmacotherapeutic group: Clostridium vaccine (cattle) Vaccine to stimulate active immunity against blackleg disease in sheep and cattle. Onset of immunity: Significant levels of immunity cannot be expected until two weeks after the second dose of the primary vaccination course. Duration of immunity: Active immunity to blackleg disease is expected to persist for up to one year. 6 PHARMACEUTICAL PARTICULARS 6.1 List of excipients Aluminium hydroxide Thiomersal Formaldehyde TRIS Maleic Acid Sodium chloride Water for injection 6.2 Incompatibilities Do not mix with any other immunological product. 6.3 Shelf-life Shelf-life of the veterinary medicinal product as packaged for sale: 3 years Shelf-life after first opening the immediate packaging: 10 hours. 6.4 Special precautions for storage Store in a refrigerator (2°C - 8°C). Do not freeze. Protect from light. 6.5 Nature and composition of immediate packaging Carton with one low density polyethylene bottle containing 50 ml closed with a butylrubber disc (Intervet UK Ltd) or rubber stopper (Laboratorios Intervet, Salamanca) with aluminium overseal combination cap. 6.6 Special precautions for the disposal of unused veterinary medicinal products or waste materials Any unused product or waste material should be disposed of in accordance with national requirements. 7 MARKETING AUTHORISATION HOLDER Intervet Ireland Ltd. Magna Drive Magna Business Park Citywest Road Dublin 24 8 MARKETING AUTHORISATION NUMBER(S) VPA 10996/142/001 9 DATE OF THE FIRST AUTHORISATION/RENEWAL OF THE AUTHORISATION 31st August 2007 10 DATE OF REVISION OF THE TEXT January 2012
Beef